During development and regeneration, organs must attain proper shape and size. This requires a control system that coordinates the behavior of individual cells to ensure the correct structure and function of the tissue. How cells sense the overall tissue status and size, remains a largely unexplored question. Researchers at the Max Planck Institute of Molecular Cell Biology and Genetics in Dresden (MPI-CBG) and the TU Dresden studied the regeneration of the liver in the mouse and discovered a novel control mechanism whereby changes in cell structure, caused by metabolic overload, inform cells about the integrity of the whole organ through a mechano-sensory mechanism. The study is published in the journal Molecular Systems Biology.
The liver has the unique capacity to regenerate. Following tissue injury, the organ restores the original tissue mass within just a few days. Previous work in the field emphasized the importance of metabolism for liver regeneration. However, it is still poorly understood, how the organ orchestrates the regenerative response throughout scales of organization to ensure the restoration of the original mass.
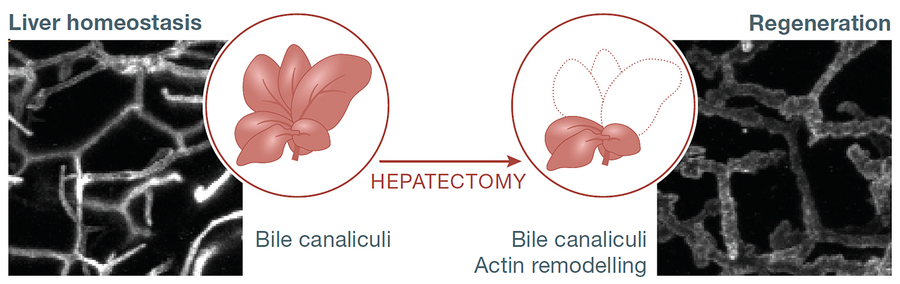
Microscopy images of the bile canaliculi network reveal structural alterations during liver regeneration (left, liver homeostasis; right, regenerating liver).
To address this problem, the research team around MPI-CBG director Marino Zerial and colleagues from the Center for Information Services and High Performance Computing (ZIH) at the TU Dresden, studied liver regeneration in the mouse. The scientists digitally reconstructed liver tissue using high‐resolution microscopy and quantitative 3D image analysis to explore tissue and cellular changes during liver regeneration. Following liver resection, the remaining tissue experiences a metabolic overload of bile acids due to the recirculation of bile acids in the body. Such overload induces an expansion of the bile canalicular network, a system of little tubings made by the liver cells (hepatocytes) to produce and distribute the bile that helps the digestion in the intestine. Remarkably, the bile canaliculi network, which is built by the plasma membrane of individual hepatocytes, senses the overload of bile acids and relays the information to the cells to induce a regenerative response. Kirstin Meyer, the first author of the study, explains: “This simple mechanism relays tissue level information on liver function to individual cells through the mechano-sensory function of the Hippo signaling pathway.” Bile acid levels are known to change not only during liver regeneration but also in response to other environmental stimuli, such as diet and circadian rhythm. Why do these changes not induce a regenerative response? Lutz Brusch (ZIH) states: “We developed a biophysical-biochemical model of bile pressure and Hippo signaling. This model suggests a mechanism that activates YAP, the effector of the Hippo pathway, in a switch-like manner.” Kirstin Meyer continues: “The threshold for activation is not reached by moderate bile acid fluctuations, but only upon severe perturbations such as tissue resection. We suggest that the bile canaliculi network acts as a self‐regulatory system that responds to critical levels of bile acids to induce the regeneration process of the liver.”
How organs control growth and regenerate, are critical biomedical questions. New insights may provide both a better understanding of abnormal growth conditions, such as cancer, and new avenues for regenerative therapies. Marino Zerial, who oversaw the study summarizes: “Our findings connect previous knowledge on the importance of bile acid metabolism for liver regeneration with the cell biology of liver cells and the organization of cells into tissues, paving the way towards a systems understanding of how cells sense instructive signals in liver regeneration.”








